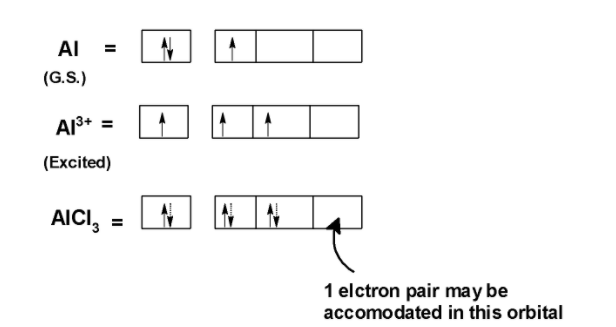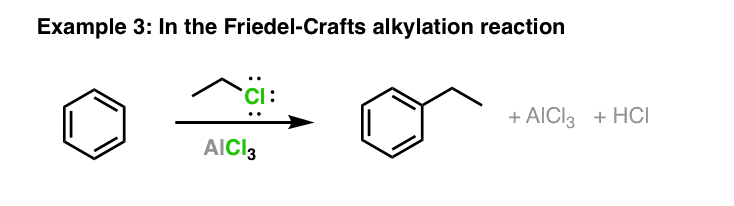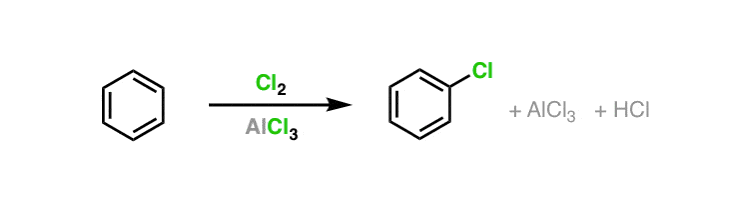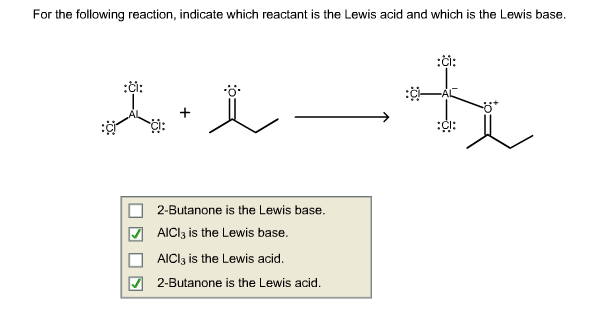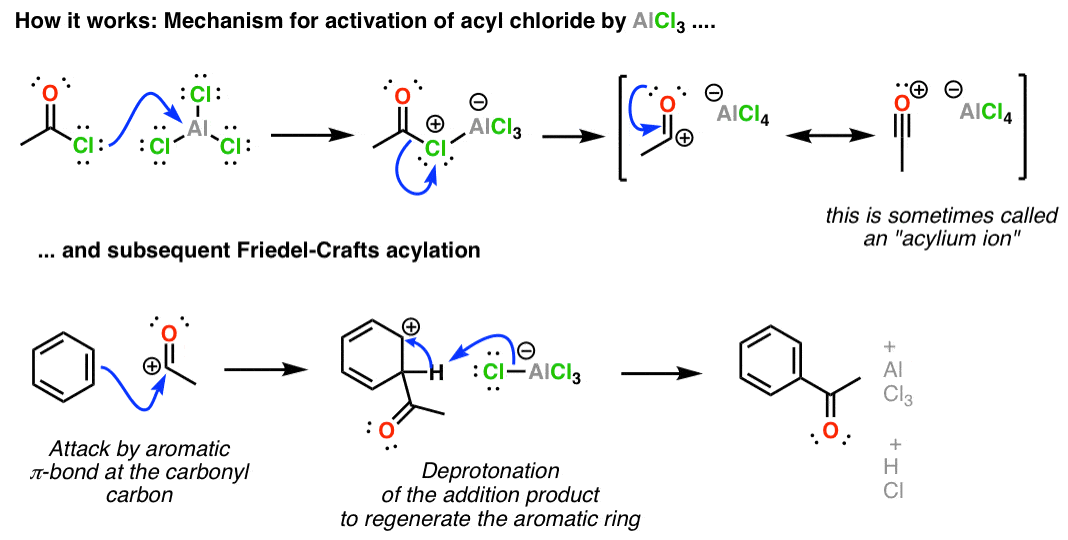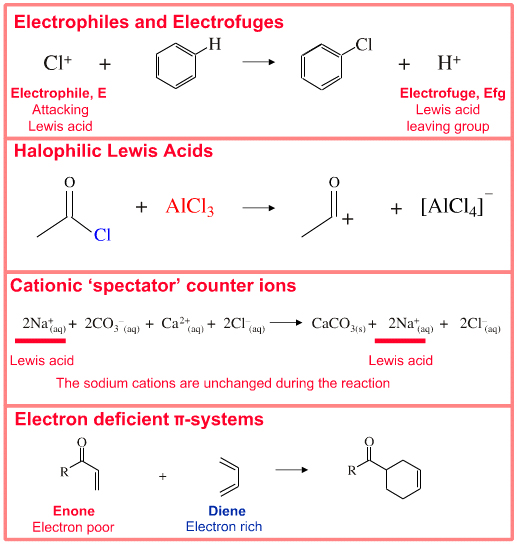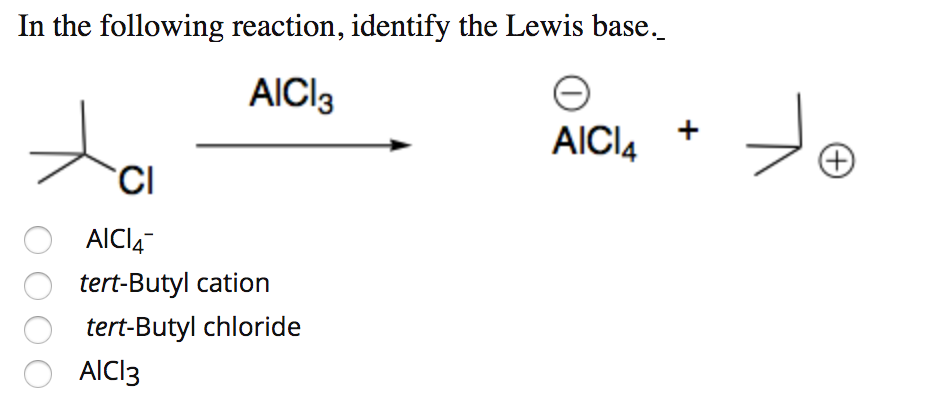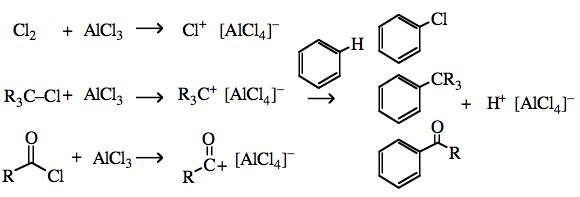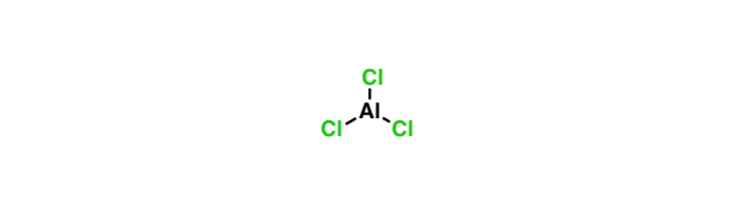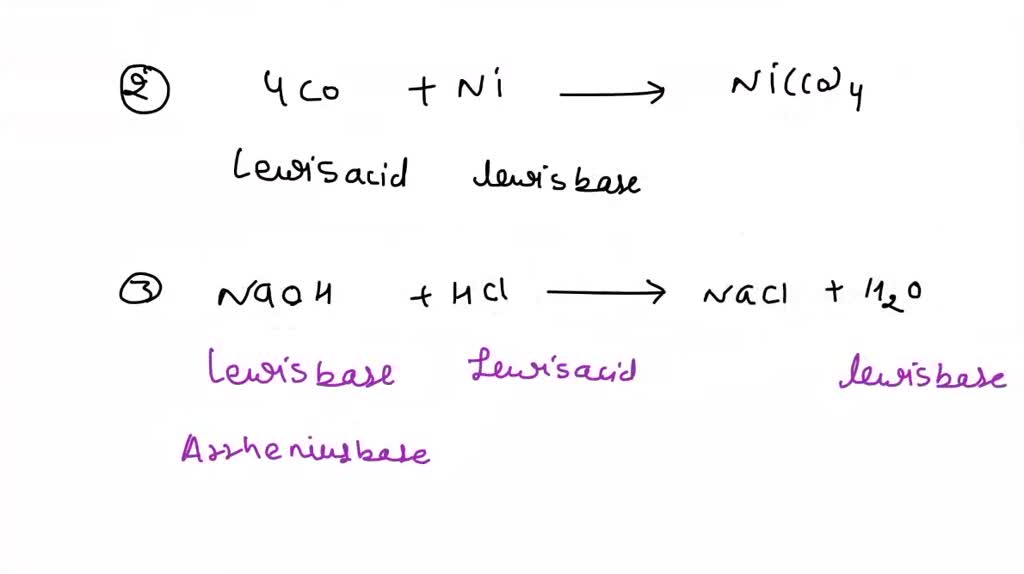
SOLVED: Circle the Lewis Acids and put a box around the Lewis bases. Na+ NH3 BF3 Pt2+ AlCl3 Fe2+ CN- H2O Ag+ H2S H+ CH3COO- Identify the Lewis acid and Lewis base

Diverging Pathways in the Activation of Allenes with Lewis Acids and Bases: Addition, 1,2-Carboboration, and Cyclization | Organometallics

AQA A Level chemistry - A2 Unit 5: Section 3.5.5 Reactions of Inorganic Compounds in Aqueous Solution - Lewis acids and bases