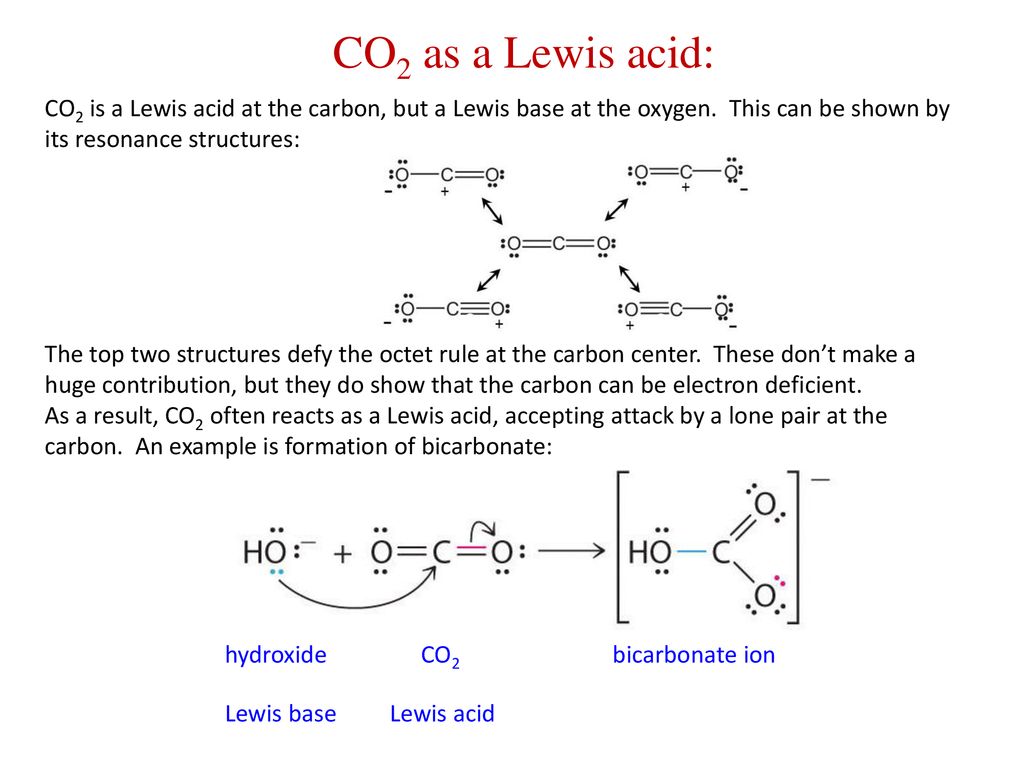
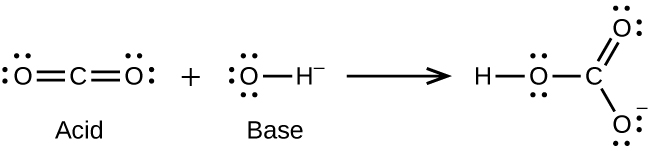
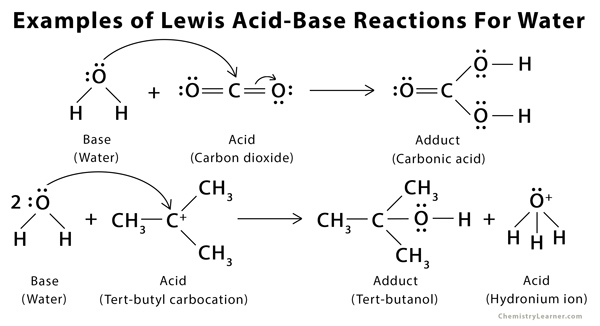
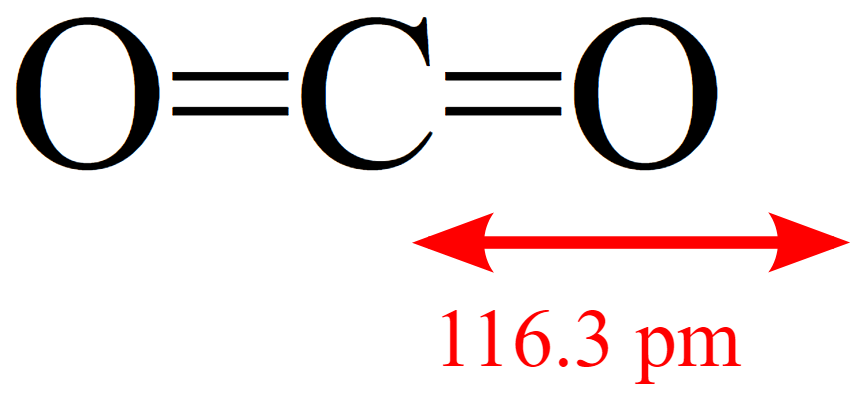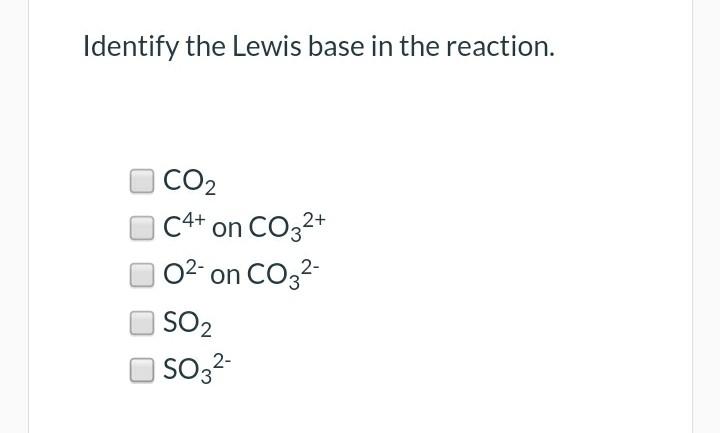Role of the Surface Lewis Acid and Base Sites in the Adsorption of CO2 on Titania Nanotubes and Platinized Titania Nanotubes: An in Situ FT-IR Study | The Journal of Physical Chemistry

Identify the lewis acid and the lewis base in the following reactions. CaO+ CO2 to CaCO3 (ii) CH3-O-CH3+AlCl3 to

Lewis Acid Strength of Interfacial Metal Sites Drives CH3OH Selectivity and Formation Rates on Cu‐Based CO2 Hydrogenation Catalysts - Noh - 2021 - Angewandte Chemie International Edition - Wiley Online Library
Minerals | Free Full-Text | CO2 Dipole Moment: A Simple Model and Its Implications for CO2-Rock Interactions

Identify the lewis acid and the lewis base in the following reactions. CaO+ CO2 to CaCO3 (ii) CH3-O-CH3+AlCl3 to

Lewis Acid Enhancement of Proton Induced CO2 Cleavage: Bond Weakening and Ligand Residence Time Effects | Journal of the American Chemical Society

Role of the Surface Lewis Acid and Base Sites in the Adsorption of CO2 on Titania Nanotubes and Platinized Titania Nanotubes: An in Situ FT-IR Study | The Journal of Physical Chemistry